How To Hold A Needle When Drawing Blood By Yourself
Blood COLLECTION:
ROUTINE VENIPUNCTURE AND SPECIMEN HANDLING
Objectives for the tutorial:
-
Describe and perform the venipuncture procedure including:
-
Appropriate vesture and protective equipment
-
Ensuring the comfort of the patient
-
Proper patient identification procedures.
-
Proper equipment option and use.
-
Proper labeling procedures and completion of laboratory requisitions.
-
Social club of draw for multiple tube phlebotomy.
-
Preferred venous access sites, and factors to consider in site selection, and ability to differentiate between the experience of a vein, tendon and artery.
-
Patient intendance following completion of venipuncture.
-
Safety and infection control procedures.
-
Quality assurance problems.
-
-
Identify the additive, additive part, volume, and specimen considerations to exist followed for each of the various color coded tubes.
-
List six areas to be avoided when performing venipuncture and the reasons for the restrictions.
-
Summarize the problems that may be encountered in accessing a vein, including the process to follow when a specimen is not obtained.
-
List several effects of exercise, posture, and tourniquet application upon laboratory values.
VENIPUNCTURE Procedure
-
The venipuncture procedure is circuitous, requiring both knowledge and skill to perform. Each phlebotomist generally establishes a routine that is comfortable for her or him.
Phlebotomists are considered to have occupational exposure to blood borne pathogens. The performance of routine vascular access procedures by skilled phlebotomists requires, at a minimum, the use of gloves to forbid contact with blood. Laboratory coats or piece of work smocks are not typically needed as personal protective equipment during routine venipuncture, but an employer must assess the workplace to determine whether certain tasks, workplace situations, or employee skill levels may event in an employee'south need for laboratory coats or other personal protective equipment to prevent contact with blood. It is an employer's responsibleness to provide, clean, repair, supercede, and/or dispose of personal protective equipment/habiliment. Equally part of presenting a professional appearance, an institutional dress lawmaking may include wearing of a laboratory coat or smock.
Several essential steps are required for every successful collection procedure:
-
Patient comfort. Is the seating comfortable and has the patient been seated for at least v minutes to avoid being rushed or confused?
-
Carry out hand hygiene earlier and subsequently each patient procedure, before putting on and after removing gloves.
-
Place the patient using two different identifiers, asking open concluded questions such as, "What is your proper noun?" and "What is your engagement of nascency?"
-
Assess the patient's physical disposition (i.eastward. nutrition, exercise, stress, basal country).
-
Check the requisition course for requested tests, patient information, and whatever special requirements.
-
Characterization the collection tubes at the bedside or drawing area.
-
Select a suitable site for venipuncture.
-
Ready the equipment, the patient and the puncture site.
-
Perform the venipuncture, collecting the sample(s) in the appropriate container(s).
-
Recognize complications associated with the phlebotomy process.
-
Assess the need for sample recollection and/or rejection.
-
Promptly send the specimens with the requisition to the laboratory.
-
ORDER Form / REQUISITION
-
A requisition course must accompany each sample submitted to the laboratory. This requisition grade must comprise the proper information in order to procedure the specimen. The essential elements of the requisition form are:
-
Patient'south surname, showtime name, and eye initial.
-
Patient'due south ID number.
-
Patient'southward engagement of birth and sexual practice.
-
Requesting doc's complete name.
-
Source of specimen. This information must be given when requesting microbiology, cytology, fluid analysis, or other testing where assay and reporting is site specific.
-
Date and time of collection.
-
Initials of phlebotomist.
-
Indicating the test(s) requested.
-
An example of a simple requisition course with the essential elements is shown below:

LABELING THE SAMPLE
-
A properly labeled sample is essential and then that the results of the exam friction match the patient. The key elements in labeling are:
-
Patient's surname, starting time and middle.
-
Patient's ID number.
-
Notation: Both of the in a higher place MUST match the same on the requisition form.
-
Date, time and initials of the phlebotomist must exist on the label of EACH tube.
-
-
Automated systems may include labels with bar codes.
-
Examples of labeled drove tubes are shown beneath:


EQUIPMENT:
-
THE FOLLOWING ARE NEEDED FOR ROUTINE VENIPUNCTURE:
-
Evacuated Collection Tubes - The tubes are designed to fill up with a predetermined volume of claret by vacuum. The prophylactic stoppers are color coded according to the additive that the tube contains. Diverse sizes are available. Blood should NEVER be poured from one tube to another since the tubes can accept dissimilar additives or coatings (run into illustrations at finish).
-
Needles - The gauge number indicates the bore size: the larger the judge number, the smaller the needle bore. Needles are available for evacuated systems and for utilise with a syringe, single draw or butterfly system.
-
Holder/Adapter - use with the evacuated collection system.
-
Tourniquet - Wipe off with alcohol and replace frequently.
-
Alcohol Wipes - seventy% isopropyl alcohol.
-
Povidone-iodine wipes/swabs - Used if blood culture is to be fatigued.
-
Gauze sponges - for application on the site from which the needle is withdrawn.
-
Adhesive bandages / record - protects the venipuncture site after collection.
-
Needle disposal unit - needles should NEVER be cleaved, aptitude, or recapped. Needles should be placed in a proper disposal unit of measurement IMMEDIATELY later their apply.
-
Gloves - can be made of latex, safe, vinyl, etc.; worn to protect the patient and the phlebotomist.
-
Syringes - may exist used in place of the evacuated collection tube for special circumstances.
Club OF DRAW
-
Blood drove tubes must be drawn in a specific club to avert cross-contagion of additives betwixt tubes. The recommended order of draw for plastic collection tubes is:
-
First - blood culture bottle or tube (yellowish or yellow-black top)
-
Second - coagulation tube (light blue top). If simply a routine coagulation assay is the just test ordered, then a unmarried calorie-free blueish top tube may be drawn. If there is a concern regarding contamination past tissue fluids or thromboplastins, and so one may draw a non-additive tube first, and then the calorie-free bluish acme tube.
-
Third - non-additive tube (red height)
-
Last depict - condiment tubes in this order:
-
SST (red-gray or gold top). Contains a gel separator and clot activator.
-
Sodium heparin (dark green peak)
-
PST (lite dark-green top). Contains lithium heparin anticoagulant and a gel separator.
-
EDTA (lavender top)
-
ACDA or ACDB (stake xanthous height). Contains acrid citrate dextrose.
-
Oxalate/fluoride (light greyness acme)
-
-
NOTE:Tubes with additives must exist thoroughly mixed. Erroneous test results may be obtained when the blood is not thoroughly mixed with the additive.
PROCEDURAL ISSUES
-
PATIENT RELATIONS AND IDENTIFICATION:
-
The phlebotomist'southward role requires a professional person, courteous, and understanding manner in all contacts with the patient. Greet the patient and identify yourself and point the procedure that will have place. Effective communication - both verbal and nonverbal - is essential.
-
Proper patient identification MANDATORY. If an inpatient is able to respond, ask for a full proper noun and e'er cheque the armband or bracelet for confirmation. DO NOT Depict BLOOD IF THE ARMBAND OR BRACELET IS MISSING. For an inpatient the nursing staff can be contacted to help in identification prior to proceeding.
-
An outpatient must provide identification other than the verbal statement of a proper noun. Using the requisition for reference, ask a patient to provide additional information such as a surname or birthdate. A regime issued photograph identification carte du jour such as a driver's license can aid in resolving identification issues.
-
If possible, speak with the patient during the process. The patient who is at ease will exist less focused on the procedure. Always give thanks the patient and excuse yourself courteously when finished.
-
PATIENT'Due south BILL OF RIGHTS:
-
The Patient'due south Nib of Rights has been adopted by many hospitals as alleged by the Articulation Committee on Accreditation of Healthcare Organizations (JCAHO). The basic patient rights endorsed by the JCAHO follow in condensed form are given below.
-
The patient has the correct to:
-
Impartial access to treatment or accommodations that are bachelor or medically indicated, regardless of race, creed, sex, national origin, or sources of payment for care.
-
Considerate, respectful care.
-
Confidentiality of all communications and other records pertaining to the patient'due south care.
-
Expect that whatever discussion or consultation involving the patient'southward case will be conducted discretely and that individuals not directly involved in the instance volition non be nowadays without patient permission.
-
Expect reasonable safety congruent with the hospital practices and environment.
-
Know the identity and professional person condition of individuals providing service and to know which physician or other practitioner is primarily responsible for his or her care.
-
Obtain from the practitioner complete and current data well-nigh diagnosis, handling, and any known prognosis, in terms the patient can reasonably be expected to sympathise.
-
Reasonable informed participation in decisions involving the patient's health care. The patient shall be informed if the hospital proposes to engage in or perform human experimentation or other inquiry/educational profits affecting his or her care or treatment. The patient has the right to decline participation in such activity.
-
Consult a specialist at the patient'due south own request and expense.
-
Refuse treatment to the extent permitted by police.
-
Regardless of the source of payment, request and receive an itemized and detailed caption of the total neb for services rendered in the infirmary.
-
Be informed of the infirmary rules and regulations regarding patient bear.
-
-
VENIPUNCTURE SITE SELECTION:
-
Although the larger and fuller median cubital and cephalic veins of the arm are used nearly frequently, the basilic vein on the dorsum of the arm or dorsal mitt veins are also acceptable for venipuncture. Foot veins are a last resort because of the higher probability of complications.
-
Certain areas are to be avoided when choosing a site:
-
Extensive scars from burns and surgery - it is difficult to puncture the scar tissue and obtain a specimen.
-
The upper extremity on the side of a previous mastectomy - exam results may be afflicted considering of lymphedema.
-
Hematoma - may cause erroneous test results. If another site is not available, collect the specimen distal to the hematoma.
-
Intravenous therapy (4) / blood transfusions - fluid may dilute the specimen, so collect from the opposite arm if possible. Otherwise, satisfactory samples may exist drawn below the Four by following these procedures:
-
Plow off the Iv for at least ii minutes before venipuncture.
-
Apply the tourniquet beneath the IV site. Select a vein other than the one with the IV.
-
Perform the venipuncture. Draw 5 ml of blood and discard before drawing the specimen tubes for testing.
-
-
Lines - Drawing from an intravenous line may avert a difficult venipuncture, simply introduces problems. The line must be flushed get-go. When using a syringe inserted into the line, blood must be withdrawn slowly to avoid hemolysis.
-
Cannula/fistula/heparin lock - hospitals accept special policies regarding these devices. In general, claret should not be fatigued from an arm with a fistula or cannula without consulting the attending physician.
-
Edematous extremities - tissue fluid aggregating alters test results.
-
-
PROCEDURE FOR VEIN SELECTION:
-
Palpate and trace the path of veins with the index finger. Arteries pulsate, are most elastic, and have a thick wall. Thrombosed veins lack resilience, feel cord-like, and ringlet easily.
-
If superficial veins are not readily apparent, you can force blood into the vein by massaging the arm from wrist to elbow, tap the site with alphabetize and 2d finger, apply a warm, damp washcloth to the site for 5 minutes, or lower the extremity over the bedside to allow the veins to fill.
-
-
PERFORMANCE OF A VENIPUNCTURE:
-
Arroyo the patient in a friendly, calm manner. Provide for their comfort as much as possible, and gain the patient's cooperation.
-
Identify the patient correctly.
-
Properly fill out advisable requisition forms, indicating the test(southward) ordered.
-
Verify the patient'southward condition. Fasting, dietary restrictions, medications, timing, and medical treatment are all of concern and should be noted on the lab requisition.
-
Cheque for any allergies to antiseptics, adhesives, or latex by observing for armbands and/or by asking the patient.
-
Position the patient. The patient should either sit in a chair, lie downward or sit up in bed. Hyperextend the patient'due south arm.
-
Apply the tourniquet 3-4 inches in a higher place the selected puncture site. Do not identify too tightly or leave on more than ii minutes (and no more than a minute to avoid increasing risk for hemoconcentration). Wait 2 minutes before reapplying the tourniquet.
-
The patient should make a fist without pumping the hand.
-
Select the venipuncture site.
-
Prepare the patient'south arm using an booze prep. Cleanse in a circular fashion, kickoff at the site and working outward. Allow to air dry.
-
Grasp the patient's arm firmly using your thumb to describe the pare taut and anchor the vein. The needle should form a 15 to 30 caste angle with the surface of the arm. Swiftly insert the needle through the skin and into the lumen of the vein. Avoid trauma and excessive probing.
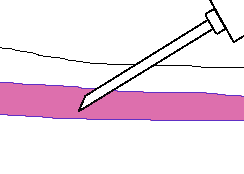
-
When the last tube to be drawn is filling, remove the tourniquet.
-
Remove the needle from the patient's arm using a swift backward motion.
-
Printing down on the gauze one time the needle is out of the arm, applying adequate pressure to avoid formation of a hematoma.
-
Dispose of contaminated materials/supplies in designated containers.
-
Mix and characterization all advisable tubes at the patient bedside.
-
Deliver specimens promptly to the laboratory.
-
PHLEBOTOMY Procedure ILLUSTRATED:
-
Patient identification
-
Filling out the requisition
-
Equipment
-
Apply tourniquet and palpate for vein
-
Sterilize the site
-
Insert needle
-
Drawing the specimen
-
Cartoon the specimen
-
Releasing the tourniquet
-
Applying pressure over the vein
-
Applying cast
-
Disposing needle into sharps
-
labeling the specimens
-
Operation OF A FINGERSTICK:
-
Follow the procedure equally outlined above for greeting and identifying the patient. As always, properly fill out appropriate requisition forms, indicating the test(s) ordered.
-
Verify the patient's condition. Fasting, dietary restrictions, medications, timing, and medical treatment are all of business concern and should be noted on the lab requisition.
-
Position the patient. The patient should either sit in a chair, lie down or sit up in bed. Hyperextend the patient'south arm.
-
The best locations for fingersticks are the 3rd (middle) and 4th (ring) fingers of the non-ascendant mitt. Do not use the tip of the finger or the center of the finger. Avoid the side of the finger where in that location is less soft tissue, where vessels and fretfulness are located, and where the bone is closer to the surface. The second (index) finger tends to have thicker, callused skin. The 5th finger tends to take less soft tissue overlying the bone. Avert puncturing a finger that is cold or cyanotic, swollen, scarred, or covered with a rash.
-
Using a sterile lancet, make a skin puncture merely off the heart of the finger pad. The puncture should exist made perpendicular to the ridges of the fingerprint and so that the drop of blood does not run down the ridges.
-
Wipe away the offset drop of blood, which tends to contain backlog tissue fluid.
-
Collect drops of blood into the collection device past gently massaging the finger. Avert excessive force per unit area that may squeeze tissue fluid into the drop of claret.
-
Cap, rotate and invert the collection device to mix the blood collected.
-
Take the patient hold a small gauze pad over the puncture site for a couple of minutes to stop the bleeding.
-
Dispose of contaminated materials/supplies in designated containers.
-
Label all advisable tubes at the patient bedside.
-
Deliver specimens promptly to the laboratory.
-
-
FINGERSTICK PROCEDURE ILLUSTRATED:
-
Equipment
-
Proper location on finger
-
Puncture with lancet
-
Drib of blood
-
Wipe commencement drop
-
Collecting the specimen
-
Specimen container
-
ADDITIONAL CONSIDERATIONS:
-
To foreclose a hematoma:
-
Puncture but the uppermost wall of the vein
-
Remove the tourniquet before removing the needle
-
Use the major superficial veins
-
Brand sure the needle fully penetrates the upper most wall of the vein. (Fractional penetration may allow blood to leak into the soft tissue surrounding the vein by way of the needle bevel)
-
Use pressure to the venipuncture site
-
-
To preclude hemolysis (which tin interfere with many tests):
-
Mix tubes with anticoagulant additives gently 5-10 times
-
Avoid drawing claret from a hematoma
-
Avoid drawing the plunger dorsum besides forcefully, if using a needle and syringe, or too small-scale a needle, and avoid frothing of the sample
-
Make sure the venipuncture site is dry out
-
Avoid a probing, traumatic venipuncture
-
Avoid prolonged tourniquet application or fist clenching.
-
-
Indwelling Lines or Catheters:
-
Potential source of exam error
-
Most lines are flushed with a solution of heparin to reduce the risk of thrombosis
-
Discard a sample at least iii times the book of the line earlier a specimen is obtained for analysis
-
-
Hemoconcentration: An increased concentration of larger molecules and formed elements in the claret may exist due to several factors:
-
Prolonged tourniquet application (no more than i minute)
-
Massaging, squeezing, or probing a site
-
Long-term IV therapy
-
Sclerosed or occluded veins
-
-
Prolonged Tourniquet Application:
-
Primary effect is hemoconcentration of non-filterable elements (i.east. proteins). The hydrostatic pressure causes some water and filterable elements to leave the extracellular space.
-
Significant increases tin be found in total protein, aspartate aminotransferase (AST), total lipids, cholesterol, iron
-
Affects packed jail cell volume and other cellular elements
-
Hemolysis may occur, with pseudohyperkalemia.
-
-
Patient Preparation Factors:
-
Therapeutic Drug Monitoring: different pharmacologic agents take patterns of administration, trunk distribution, metabolism, and elimination that affect the drug concentration as measured in the blood. Many drugs will have "peak" and "trough" levels that vary according to dosage levels and intervals. Check for timing instructions for drawing the appropriate samples.
-
Effects of Exercise: Muscular activeness has both transient and longer lasting furnishings. The creatine kinase (CK), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), and platelet count may increment.
-
Stress: May cause transient elevation in white blood cells (WBC's) and elevated adrenal hormone values (cortisol and catecholamines). Anxiety that results in hyperventilation may cause acid-base imbalances, and increased lactate.
-
Diurnal Rhythms: Diurnal rhythms are body fluid and analyte fluctuations during the twenty-four hours. For example, serum cortisol levels are highest in early morn only are decreased in the afternoon. Serum iron levels tend to driblet during the 24-hour interval. You must check the timing of these variations for the desired drove point.
-
Posture: Postural changes (supine to sitting etc.) are known to vary lab results of some analytes. Certain larger molecules are non filterable into the tissue, therefore they are more concentrated in the claret. Enzymes, proteins, lipids, iron, and calcium are significantly increased with changes in position.
-
Other Factors: Age, gender, and pregnancy have an influence on laboratory testing. Normal reference ranges are oft noted according to age.
-
REASONS FOR CANCELING A LABORATORY TEST
-
A test that has been ordered may be cancelled due to bug unrelated to drawing the specimen, and these are the most common causes for cancellations:
-
Duplicate examination asking
-
Wrong test ordered
-
Test no longer needed
-
-
A test may be cancelled due to a technical problem in the specimen collection process:
-
Hemolysis of the specimen
-
Clotted specimen
-
Quantity of specimen not sufficient
-
Collection of specimen in incorrect tube
-
Contaminated specimen
-
Identification of the specimen is doubtable
-
Delay in send - specimen too old
-
Condom AND INFECTION CONTROL
-
Because of contacts with ill patients and their specimens, it is of import to follow rubber and infection control procedures.
-
PROTECT YOURSELF
-
Practice universal precautions:
-
Wear gloves and a lab coat or gown when treatment blood/body fluids.
-
Alter gloves later on each patient or when contaminated.
-
Launder easily frequently.
-
Dispose of items in advisable containers.
-
-
Dispose of needles immediately upon removal from the patient's vein. Do not bend, break, recap, or resheath needles to avoid adventitious needle puncture or splashing of contents.
-
Clean upward any blood spills with a disinfectant such as freshly made 10% bleach.
-
If yous stick yourself with a contaminated needle:
-
Remove your gloves and dispose of them properly.
-
Squeeze puncture site to promote bleeding.
-
Wash the surface area well with soap and water.
-
Record the patient'south proper name and ID number.
-
Follow institution's guidelines regarding treatment and follow-up.
-
NOTE: The utilise of safety zidovudine following blood exposure to HIV has shown effectiveness (virtually 79%) in preventing seroconversion
-
-
-
PROTECT THE PATIENT
-
Identify blood collection equipment away from patients, especially children and psychiatric patients.
-
Practice hygiene for the patient's protection. When wearing gloves, alter them between each patient and wash your easily frequently. Always vesture a clean lab glaze or gown.
-
TROUBLESHOOTING GUIDELINES:
-
IF AN INCOMPLETE Collection OR NO BLOOD IS OBTAINED:
-
Change the position of the needle. Movement it forrad (it may not be in the lumen)

-
or move it astern (it may take penetrated too far).

-
Adjust the angle (the bevel may exist against the vein wall).

-
Loosen the tourniquet. It may be obstructing blood flow.
-
Try another tube. Utilize a smaller tube with less vacuum. At that place may be no vacuum in the tube beingness used.
-
Re-anchor the vein. Veins sometimes curlicue away from the point of the needle and puncture site.
-
Accept the patient make a fist and flex the arm, which helps engorge muscles to fill up veins.
-
Pre-warm the region of the vein to reduce vasoconstriction and increase blood period.
-
Have the patient drink fluids if dehydrated.
-
-
IF Blood STOPS FLOWING INTO THE TUBE:
-
The vein may have complanate; resecure the tourniquet to increment venous filling. If this is not successful, remove the needle, take care of the puncture site, and redraw.

-
The needle may accept pulled out of the vein when switching tubes. Hold equipment firmly and place fingers confronting patient's arm, using the flange for leverage when withdrawing and inserting tubes.
-
-
Issues OTHER THAN AN INCOMPLETE Drove:
-
A hematoma forms under the skin adjacent to the puncture site - release the tourniquet immediately and withdraw the needle. Apply firm pressure.
Hematoma formation is a problem in older patients.

-
The blood is vivid carmine (arterial) rather than venous. Utilize firm force per unit area for more than 5 minutes.

-
BLOOD Collection ON BABIES:
-
The recommended location for claret collection on a newborn baby or infant is the heel. The diagram beneath indicates in green the proper area to use for heel punctures for blood collection:
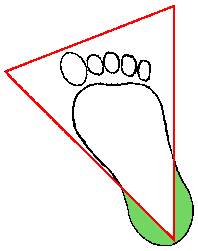
-
Prewarming the infant's heel (42 C for 3 to 5 minutes) is of import to obtain capillary blood gas samples and warming also greatly increases the flow of claret for drove of other specimens. However, exercise not use as well high a temperature warmer, because baby's skin is thin and susceptible to thermal injury.
-
Clean the site to be punctured with an alcohol sponge. Dry out the cleaned area with a dry cotton fiber sponge. Concord the baby's foot firmly to avoid sudden movement.
-
Using a sterile blood lancet, puncture the side of the heel in the appropriate regions shown above in green. Exercise not use the fundamental portion of the heel because you might injure the underlying bone, which is close to the skin surface. Practise non apply a previous puncture site. Make the cut beyond the heelprint lines and then that a driblet of blood tin can well up and not run down along the lines.
-
Wipe away the first drop of claret with a piece of make clean, dry cotton fiber. Since newborns do not frequently bleed immediately, utilise gentle force per unit area to produce a rounded drop of blood. Exercise not utilize excessive pressure or heavy massaging because the claret may become diluted with tissue fluid.
-
Make full the capillary tube(s) or micro collection device(s) as needed.
-
When finished, drag the heel, place a piece of clean, dry cotton wool on the puncture site, and concur it in place until the bleeding has stopped.
-
Exist sure to dispose of the lancet in the appropriate sharps container. Dispose of contaminated materials in appropriate waste receptacles. Remove your gloves and wash your easily.
-
HEELSTICK Procedure ILLUSTRATED:
-
Heelstick on baby
-
PEDIATRIC PHLEBOTOMY:
-
Children, particularly nether the age of 10, may experience hurting and anxiety during the phlebotomy procedure.
-
A multifariousness of techniques can be employed to reduce pain and feet. Constructive methods use distraction. These may include listening to music or a story, watching a video, playing with a toy, having a parent provide distraction with talk or affect, using wink cards, and squeezing a rubber ball. (Uman et al, 2013)
Collection TUBES FOR PHLEBOTOMY
-
Collection tubes can vary in size for volume of blood drawn, appropriate to the tests ordered with sample size required, and vary in the kind of additive for anticoagulation, separation of plasma, or preservation of analyte. Larger tube sizes typically provide for collection of samples from half dozen to 10 mL.

-
Smaller collection tubes for sample sizes of ii mL or less may be appropriate in situations where a smaller amount claret should be fatigued, every bit in pediatric patients, or to minimize hemolysis during collection, or to avoid insufficient sample volume in the collection tube.

| Red Summit |  |
|---|---|
| Condiment | None |
| Style OF ACTION | Blood clots, and the serum is separated by centrifugation |
| USES | Chemistries, Immunology and Serology, Blood Bank (Crossmatch) |
| Gold Elevation |  |
|---|---|
| ADDITIVE | None |
| Fashion OF Activity | Serum separator tube (SST) contains a gel at the bottom to dissever blood from serum on centrifugation |
| USES | Chemistries, Immunology and Serology |
| Light Green Top |  |
|---|---|
| ADDITIVE | Plasma Separating Tube (PST) with Lithium heparin |
| MODE OF Action | Anticoagulates with lithium heparin; Plasma is separated with PST gel at the bottom of the tube |
| USES | Chemistries |
| Imperial Top |  |
|---|---|
| ADDITIVE | EDTA |
| MODE OF ACTION | Forms calcium salts to remove calcium |
| USES | Hematology (CBC) and Claret Banking concern (Crossmatch); requires full depict - invert viii times to prevent clotting and platelet clumping |
| Light Blue Top |  |
|---|---|
| Additive | Sodium citrate |
| Fashion OF ACTION | Forms calcium salts to remove calcium |
| USES | Coagulation tests (protime and prothrombin fourth dimension), full draw required |
| Green Height |  |
|---|---|
| Additive | Sodium heparin or lithium heparin |
| Manner OF ACTION | Inactivates thrombin and thromboplastin |
| USES | For lithium level, use sodium heparin For ammonia level, use sodium or lithium heparin |
| Dark Blue Top |  |
|---|---|
| ADDITIVE | EDTA- |
| Fashion OF ACTION | Tube is designed to contain no contaminating metals |
| USES | Trace chemical element testing (zinc, copper, lead, mercury) and toxicology |
| Light Gray Top |  |
|---|---|
| ADDITIVE | Sodium fluoride and potassium oxalate |
| MODE OF ACTION | Antiglycolytic agent preserves glucose up to five days |
| USES | Glucoses, requires full draw (may cause hemolysis if brusque draw) |
| Yellowish Top |  |
|---|---|
| Additive | ACD (acrid-citrate-dextrose) |
| Way OF Activity | Complement inactivation |
| USES | HLA tissue typing, paternity testing, DNA studies |
| Yellow - Black Top |  |
|---|---|
| ADDITIVE | Goop mixture |
| Fashion OF ACTION | Preserves viability of microorganisms |
| USES | Microbiology - aerobes, anaerobes, fungi |
| Black Pinnacle |  |
|---|---|
| ADDITIVE | Sodium citrate (buffered) |
| Style OF Activeness | Forms calcium salts to remove calcium |
| USES | Westergren Sedimentation Charge per unit; requires full draw |
| Orange Top |  |
|---|---|
| ADDITIVE | Thrombin |
| Fashion OF Action | Quickly clots claret |
| USES | STAT serum chemistries |
| Light Brown Acme |  |
|---|---|
| ADDITIVE | Sodium heparin |
| Style OF ACTION | Inactivates thrombin and thromboplastin; contains nigh no lead |
| USES | Serum atomic number 82 determination |
| Pinkish Top |  |
|---|---|
| Condiment | Potassium EDTA |
| Fashion OF ACTION | Forms calcium salts |
| USES | Immunohematology |
| White Pinnacle |  |
|---|---|
| Condiment | Potassium EDTA |
| MODE OF Activity | Forms calcium salts |
| USES | Molecular/PCR and bDNA testing |
References
-
Giavarina D, Lippi G. Blood venous sample drove: Recommendations overview and a checklist to better quality. Clin Biochem. 2017;l(10-11):568-573.
Kiechle FL. So Yous're Going to Collect a Blood Specimen: An Introduction to Phlebotomy, 13th Edition (2010), College of American Pathologists, Northfield, IL.
Dalal BI, Brigden ML. Factitious biochemical measurements resulting from hematologic conditions. Am J Clin Pathol. 2009 Feb;131(2):195-204.
Lippi G, Salvagno GL, Montagnana One thousand, Franchini M, Guidi GC. Phlebotomy issues and quality improvement in results of laboratory testing. Clin Lab. 2006;52(five-half-dozen):217-30.
Lippi G, Blanckaert N, Bonini P, Dark-green Southward, Kitchen S, Palicka Five, Vassault AJ, Mattiuzzi C, Plebani M. Causes, consequences, detection, and prevention of identification errors in laboratory diagnostics. Clin Chem Lab Med. 2009;47(two):143-53.
Occupational Safety and Health Assistants, United states of america Department of Labor. https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=INTERPRETATIONS&p_id=25913 and https://www.osha.gov/OshDoc/data_BloodborneFacts/bbfact03.pdf (Accessed Jan 10, 2018).
Phelan MP, Reineks EZ, Berriochoa JP, Schold JD, Hustey FM, Chamberlin J, Kovach A. Impact of Use of Smaller Volume, Smaller Vacuum Blood Collection Tubes on Hemolysis in Emergency Department Blood Samples. Am J Clin Pathol. 2017;148(4):330-335.
Uman LS, Birnie KA, Noel Yard, et al. Psychological interventions for needle-related procedural pain and distress in children and adolescents. Cochrane Database Syst Rev. 2013 October x;(x):CD005179. doi: 10.1002/14651858.CD005179.pub3.
Valenstein PN, Sirota RL. Identification errors in pathology and laboratory medicine. Clin Lab Med. 2004;24(four):979-96, vii.
World Health Organization. WHO guidelines on cartoon blood: best practices in phlebotomy. https://world wide web.ncbi.nlm.nih.gov/books/NBK138650/pdf/Bookshelf_NBK138650.pdf (Accessed January 10, 2018)
And for our furry friends:
Joslin JO. Blood Collection Techniques in Exotic Minor Mammals. Journal of Exotic Pet Medicine. 2009;18(2):117-139.
Source: https://webpath.med.utah.edu/TUTORIAL/PHLEB/PHLEB.html
Posted by: garnergrele1962.blogspot.com


0 Response to "How To Hold A Needle When Drawing Blood By Yourself"
Post a Comment